Concrete chemistry is obviously tough to understand for a small house owner. Spending some amount towards concrete carbonation test is an excellent idea to know the effect of the atmospheric CO2 on the RCC structure. The concrete carbonation test for in-situ concrete is associated with the corrosion of reinforcement steel. Due to the concrete carbonation, reinforcement corrosion often occurs on the building facades which are exposed to moisture, rainfall and shaded from sunlight. The steel corrosion in concrete also occurs due to the carbonation, when the concrete has a least cover over the steel reinforcement.
Concrete is basically alkaline in nature having pH more than 12.6. It is due to its alkalinity that the cover of concrete protects reinforcement from corrosion. Due to concrete carbonation, its alkalinity reduces and having a way for the corrosion. The corrosion leads to an expansion in volume of film over steel bar. This expansion in volume create cracks in concrete. This cracks further invite CO2. Thus the vicious cycle continues and ultimately leading structure in a disaster state.

Courtesy - amazonaws
Also, learn other tests which are used to check the Hardened Concrete.
Rebound Hammer Test Procedure
Ultrasonic Testing Procedure
Concrete Pull Out Test
Concrete Core Test
Chloride Content Test
Here are some details on the concrete carbonation test for the analysis of the structures.
Concrete Carbonation occurs when the atmospheric carbon dioxide (CO2) reacts with hydrated cement minerals (CaOH2). CO2 react in the presence of moisture with CaOH2 and produce carbonates (CaCO3). Carbonates slowly penetrate below the exposed surface of the concrete. Thus the carbonation affects the concrete cover over the reinforcing steel. Hence steel corrosion occurs due to the process of concrete carbonation. The concrete loses its durability, and finally, it cracks due to the expansion of film over the bar. At the same time, the rate of steel corrosion is at the highest level. The concrete carbonation process is also called Concrete depassivation.
Factor Affecting Concrete Carbonation Process:
The concrete carbonation is a slow process. The concrete carbonation highly depends on humidity, temperature and atmospheric carbon dioxide CO2.
01. Relative Humidity:
The Carbonation of concrete occurs when the level of humidity in the atmosphere is ideally between 50 to 70%.
If humidity is below the normal level (<50%) then there is not enough water vapour present in the atmosphere. Hence CO2 cannot dissolve.
Furthermore, if humidity is higher (>70%), more water vapour (atmospheric moisture) is present in the atmosphere. Hence pores of the concrete are filled with water and CO2 cannot enter into the concrete. Ultimately no CO2 diffusion takes place.
The most dangerous level of humidity for carbonation is 50 to 70% at which CO2 react with calcium hydroxide (CaOH2) and generate more carbonates. The generation of carbonates in concrete is called the process of carbonation. Ultimately, carbonation process reduces the level of the concrete pH from 13 to 9.
02. Temperature:
The carbonation is worse in hot environments.
03. Atmospheric CO2:
The concentration of CO2 gas in the atmosphere is about 0.04% (400 ppm) by volume of the atmosphere, but it is increasing annually due to motor vehicles and fossil fuel burning in cities. Which is also known as “Green House Effect”.
The Procedure of Concrete Carbonation Test:
When the steel is surrounded by concrete layer, Hydrated cement provides an alkaline environment at which concrete pH is around 12.6. It helps steel to form the passive film on the surface of the steel. The passive film protects the reinforcement steel from the corrosion. Thus concrete cover provides protection to the steel.
The attack of the atmospheric CO2 reduces the alkalinity in the concrete. The concrete porosity allows the CO2 to react with the alkalis like calcium, sodium and potassium hydroxides which are formed due to cement hydration in concrete. The reaction produces the carbonates which reduce the level of concrete pH. When carbonates ingress deeply in concrete, they also break the protective passive film of the steel. So the oxygen and moisture are easily available to steel, and it gets corroded. Hence we need to perform the carbonation test to determine the level or degree of carbonation to evaluate the rate of corrosion.
Two Method Measure the Level of Concrete Carbonation:
01. IR Spectrum Analysis for Concrete Carbonation:
IR spectrum method measures the concentration of CO2 absorbed by the concrete specimen. This method of testing is not much attractive for chemical testing of concrete.
The IR spectrum method set up consists of a closed loop in which a mixture of air and carbon dioxide could be introduced at certain relative humidity. The pump circulates the mixture of air and CO2. The concentration of CO2 is reduced in gas mixture due to the carbonation reaction. The concentration of Co2 in gas is measured by using IR absorption device. The relative humidity and temperature is also recorded.
02. pH Indicator Analysis for Concrete Carbonation:
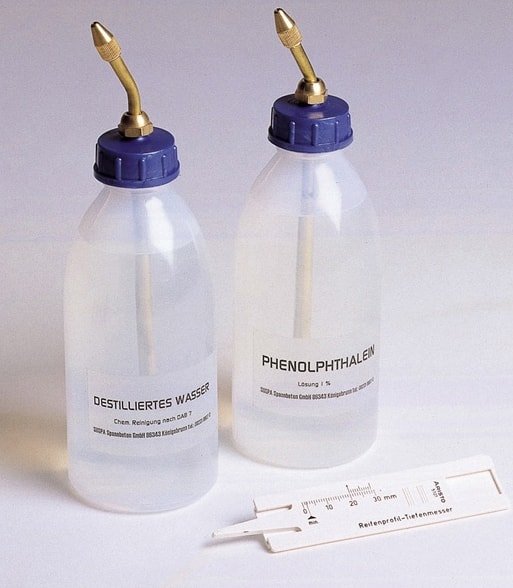
Courtesy - controls-group
The pH indicator method is a very popular method for chemical testing of concrete. A spray of 0.2% solution of phenolphthalein chemical is done on the surface of the concrete to find the area affected by the carbonation. Phenolphthalein solution indicates the change of pH level in the concrete. If the concrete changes its grey colour to pink, it means that the concrete is in good condition. When there is no change in colour of concrete, it means that the area is affected by carbonation.
The carbonation test is performed by extracting the cores of in-situ concrete. The carbonation test is also performed by drilling a hole on the concrete surface to the different depth up to concrete cover. Remove the dust by brush and Blow the air to clean the hole.
Spray 0.2% solution of the phenolphthalein with physician’s injection syringe or needle on such freshly drilled/broken concrete and observe the change in colour. Measure the depth of uncoloured layer (carbonated layer) in millimetres from the external surface at 4 to 8 positions. Take the average of measurement.
The Depth of Concrete Carbonation:

Courtesy - Researchgate
The Depth of concrete carbonation is estimated by the change in colour profile, and the degree of carbonation that can be measured in millimetre. The depth of carbonation is proportional to the square root of time. I.e, if the depth of carbonation is 1 millimetre in one-year-old concrete, it will be 3mm after 9 years, 5mm after 25 years and 10mm after 100 years.
We can also determine the pH value by analyzing the sample of core powder collected by drilling from the site. It is determined by dissolving the powder sample in distilled water and thereafter titration in the laboratory.
The phenolphthalein method for concrete carbonation test is a simple and cheap method. It easily determines the depth of carbonation in concrete. It also helps to decide about the information on the risk of reinforcement corrosion. The only limitation is the minor amount of damage done to the concrete surface by drilling a core.
In concrete carbonation test, the depth of carbonation is also confirmed with microscopy either optical microscopy using the thin concrete section or scanning electron microscopy using polished concrete section. The carbonation test is very useful to make an initial assessment of reinforced concrete structures. Carbonation test is quick, easy and used in the current scenario for the carbonation affected in-situ concrete.